|
Hemophilia affects the circulatory system, as it affects blood clotting. Blood clots are also called thrombi (singular thrombus), and the process in which a “leaky” blood vessel is repaired is called hemostasis.
|
Quick Facts
The opposite of hemostasis is hemorrhage, where blood is actively leaving the circulatory system, which will cause problems such as brain hemorrhages. Thrombosis is when a clot forms in an undamaged vessel, which can lead to strokes and heart attacks. a short video about hemophilia and excessive bleeding
|
Hemostasis: Repairing Blood Vessel Damage
1. Vascular Spasm: When a blood vessel has burst, whether due to an external cut or leaking below the surface of the skin, the smooth muscle around the burst impulsively contract to decrease the blood flow to the leaking area. This prevents excessive blood loss. After the vessels have contracted, platelets that flow through the blood start working on the tear.
1. Vascular Spasm: When a blood vessel has burst, whether due to an external cut or leaking below the surface of the skin, the smooth muscle around the burst impulsively contract to decrease the blood flow to the leaking area. This prevents excessive blood loss. After the vessels have contracted, platelets that flow through the blood start working on the tear.
|
2. Platelet Plug Formation: Platelets are small cells produced in our bone marrow, and about 30% are stored in the spleen while the rest flow through our bloodstream. These cells have jagged edges that catch on the rough snags of broken blood vessels, and help block and heal the leak. After flowing through the blood for about a week to 10 days, platelets that have not been used to heal cuts are destroyed by cells in the liver or spleen. Platelets go through 4 stages to stop bleeding: adhesion, activation and secretion, and aggregation.
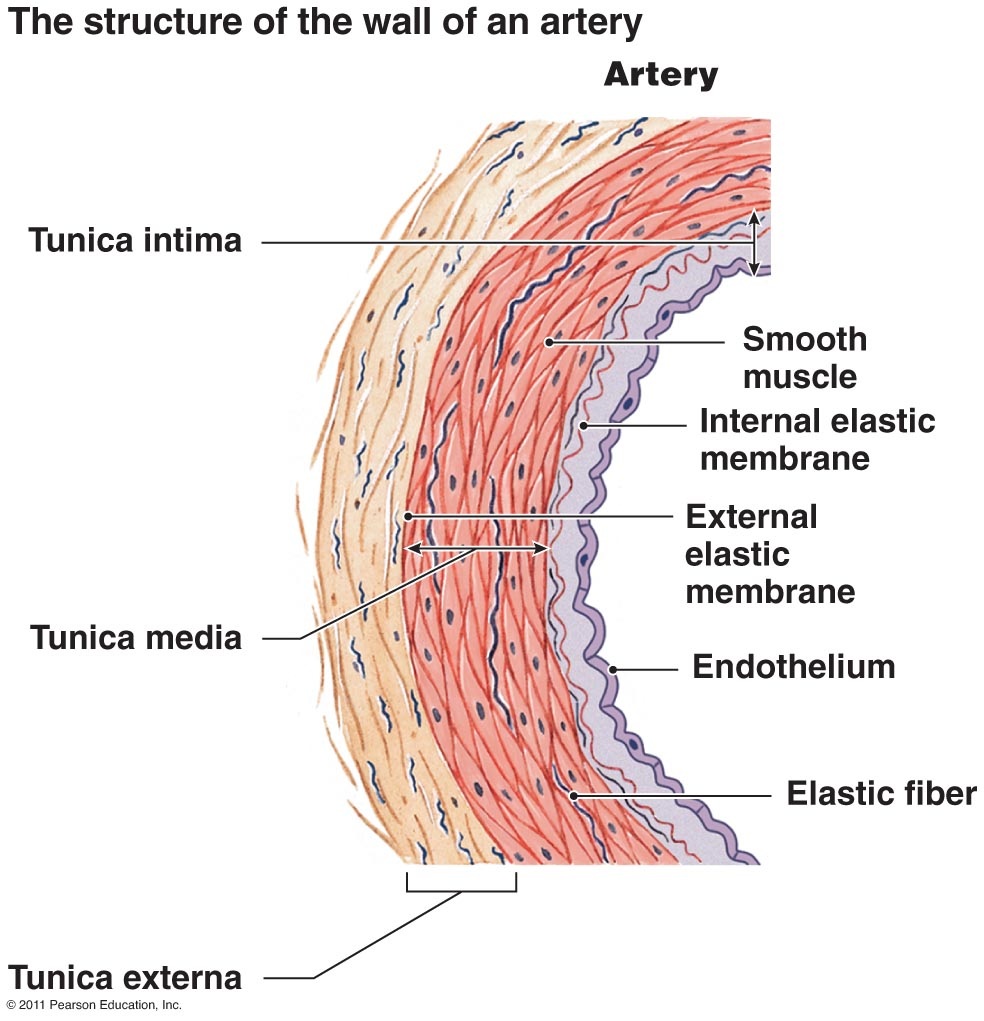
Adhesion: When a blood vessel is torn, the outer layer of the vessel, tunica externa, comes in contact with the blood. The tunica externa is made up of collagen fibres that anchor the vessel to the surrounding areas; platelets are attracted to this collagen and stick to the area. This, along with the jagged edges of the cut on the blood vessel, encourages the already-jagged platelets to adhere to the cut like velcro. Activation and Secretion: All these processes flow, but with these two stages, it’s especially hard to tell when one stage starts and the other ends. There are storage spaces inside each platelet that store chemicals called granules; when enough platelets have stuck onto each other, they start to change form and grow long ‘arms’ (activation). This causes the the granules to release chemicals that signal and attract even more platelets, to ensure a tight seal around the wound (secretion). When platelets change shapes, they have been “activated”; the stage in which they release chemicals is called “secretion”. Aggregation: When platelets stick to the blood vessel wall, it is called “adhesion”; when they stick to each other, which is necessary to ensure that a secure clot is formed, it is called “aggregation”. Platelets are only a temporary solution to a blood vessel tear, as they are not strong enough on their own. |
Below: platelets
diagram of cell layers
|
3. Coagulation: Coagulation is the process in which liquid blood solidifies, or at least turns to a semi-solid state. This is a complicated process that happens with the help of 13 main clotting factors, numbered with Roman numerals (I, II, III, IV...XIII), though there are more clotting factors beyond the 13. If blood successfully coagulates, a strong fibrin clot made up of fibrin strands will be able to cover the injury to the vessel and successfully stop blood leakage.
1. Formation of Prothrombinase
Prothrombinase starts the process to forming a fibrin clot. There are 2 ways prothrombinase can be created.
a) Extrinsic (Tissue factor) Pathway: Inactive factor VII comes in contact with the Tissue factor on the surface of the vessel (because of the break in the vessel) and becomes activated into a protease, which is an enzyme capable of breaking chemical bonds in proteins. Activated factor VII breaks the bonds in factor X, which then combines with factor V to form prothrombinase
b) Intrinsic (contact activation) Pathway: When the inactive factor XII contacts damaged tissue, it activates and becomes a protease. factor XII then breaks bonds and activates in factor XI, turning it into a protease as well, which then breaks bonds and activates factor IX, which also becomes a protease. factor IX, along with factor VIII, activates factor X which then joins factor V to form prothrombinase.
2. Prothrombinase converts Prothrombin, a plasma protein made in the liver (also known as clotting factor II), into thrombin.
3. The liver produces a protein called fibrinogen (clotting factor I) that flows in the blood that remains inactive while flowing through the blood. Chemical groups at the end of each fibrinogen molecule prevent it from becoming sticky fibrin, and prevent it from sticking onto the surfaces of healthy blood vessels (which would form a thrombus that, if large enough, can obstruct blood flow and cause tissue death). When platelets are activated, thousands of fibrinogen strands start attaching to the platelet plug. Thrombin can then remove the chemical groups at the ends of fibrinogen molecules, converting fibrinogen into the sticky fibrin, which then creates a strong clot. Thrombin also activates factor XIII, which allows fibrin strands to attach to each other as well as to platelets, which strengthens the clot considerably.
In short, when there is a defect in the cell wall, smooth muscles contract and restrict blood flow to lessen blood loss, platelets adhere to the tear, activate and secrete, and aggregate; prothrombinase is formed, which triggers a domino effect until fibrin is created and sticks to the platelet plug, creating a strong clot.
The musculoskeletal system is also affected by this disorder, because one of the symptoms is that blood pools in joints such as the knee. The pressure of the blood can damage the cartilage, which can permanently damage the joint.
The immune system sometimes reacts to replacement therapy (see Diagnosis & Treatment). In the case of acquired hemophilia, the immune system produces antibodies that attack clotting factors.
The liver is responsible for producing the clotting factors, so it is theoretically affected although all other liver functions are normal. Hemophilia can affect most other systems indirectly, such as the nervous system by causing a brain hemorrhage through excessive bleeding from a minor injury. If a bleeding episode happens around or near an organ, the organ can be affected negatively as can the body system it belongs to.
1. Formation of Prothrombinase
Prothrombinase starts the process to forming a fibrin clot. There are 2 ways prothrombinase can be created.
a) Extrinsic (Tissue factor) Pathway: Inactive factor VII comes in contact with the Tissue factor on the surface of the vessel (because of the break in the vessel) and becomes activated into a protease, which is an enzyme capable of breaking chemical bonds in proteins. Activated factor VII breaks the bonds in factor X, which then combines with factor V to form prothrombinase
b) Intrinsic (contact activation) Pathway: When the inactive factor XII contacts damaged tissue, it activates and becomes a protease. factor XII then breaks bonds and activates in factor XI, turning it into a protease as well, which then breaks bonds and activates factor IX, which also becomes a protease. factor IX, along with factor VIII, activates factor X which then joins factor V to form prothrombinase.
2. Prothrombinase converts Prothrombin, a plasma protein made in the liver (also known as clotting factor II), into thrombin.
3. The liver produces a protein called fibrinogen (clotting factor I) that flows in the blood that remains inactive while flowing through the blood. Chemical groups at the end of each fibrinogen molecule prevent it from becoming sticky fibrin, and prevent it from sticking onto the surfaces of healthy blood vessels (which would form a thrombus that, if large enough, can obstruct blood flow and cause tissue death). When platelets are activated, thousands of fibrinogen strands start attaching to the platelet plug. Thrombin can then remove the chemical groups at the ends of fibrinogen molecules, converting fibrinogen into the sticky fibrin, which then creates a strong clot. Thrombin also activates factor XIII, which allows fibrin strands to attach to each other as well as to platelets, which strengthens the clot considerably.
In short, when there is a defect in the cell wall, smooth muscles contract and restrict blood flow to lessen blood loss, platelets adhere to the tear, activate and secrete, and aggregate; prothrombinase is formed, which triggers a domino effect until fibrin is created and sticks to the platelet plug, creating a strong clot.
The musculoskeletal system is also affected by this disorder, because one of the symptoms is that blood pools in joints such as the knee. The pressure of the blood can damage the cartilage, which can permanently damage the joint.
The immune system sometimes reacts to replacement therapy (see Diagnosis & Treatment). In the case of acquired hemophilia, the immune system produces antibodies that attack clotting factors.
The liver is responsible for producing the clotting factors, so it is theoretically affected although all other liver functions are normal. Hemophilia can affect most other systems indirectly, such as the nervous system by causing a brain hemorrhage through excessive bleeding from a minor injury. If a bleeding episode happens around or near an organ, the organ can be affected negatively as can the body system it belongs to.